The Importance Of using FDA Approved Dental Materials

Considering the high costs of dental treatments if things go wrong, you ensure that the dental appliances you’re using have FDA approved dental materials in them. The standards set by FDA for dental materials ensure that they are up to the quality required to keep your dental health fine.
In this article, we’re explaining what is FDA, how it approves dental materials and the importance of using FDA approved dental materials.
What is the FDA?
The Food and Drug Administration is responsible for protecting public health by ensuring the quality, effectiveness and protection of human and veterinary medicines, biological products and medical devices; and by ensuring the safety of our nation’s food supply, cosmetics and radiation-emitting materials.
The FDA is responsible for promoting public health by helping to accelerate innovations that make medical products more efficient, safer and more affordable, and by helping the public to obtain accurate, science-based information that they need to use in order to maintain and improve their health.
The U.S. Food and Drug Administration (FDA) reviews, evaluates and oversees a wide variety of medicinal products, including medications, medical equipment, food, cosmetics and many other health related products. In the simplest words, “FDA approval” means that the FDA has determined that the benefits of the approved item outweigh its possible risks.
Until the FDA, marketers were able to make claims about a product without evidence that it was healthy or that it even worked. It has left customers particularly insecure. Now, new products must go through the FDA approval process before they are made available to the public.
Why is the approval process of the FDA important?
The approval of the FDA is essential as it validates the need for research on how medications and dental materials work for children, not just adults. It also helps one to better assess the correct dose for kids, determine the best route of administration, and check for any drug interactions.
How is a medication or device approved by the FDA?
In order to be approved by the FDA for a medication or medical device, the manufacturer must prove to the FDA that the item is “safe and successful.” While no product or medical device is entirely risk-free, the research and testing must prove that the advantages of the medication or device for a specific condition outweigh the risks to the patients using the item.
Here’s a short explanation of the steps taken by the FDA-approved drug:
Drug Developed: A company is making a new product and is seeking FDA approval of it for sale in the U.S.
Animal testing: The company will check the new drug on animals before researching the drug on humans to find out whether it has the potential to cause significant harm (i.e. toxicity).
IND Application: The firm submits to the FDA an application for Investigational New Drug (IND), based on the findings of the initial animal test. These results must include the composition and manufacturing of the drug and the proposed plan to test the drug on humans.
Clinical trials: Clinical trials to test the drug on people will start after the FDA reviews and approves the IND application. There are 4 phases of clinical trials followed by large-scale trials, starting with small-scale trials. The researchers then send details of the analysis to the FDA after the clinical trials.
NDA Application: The company may file a New Drug Application (NDA) after a drug developer has received evidence that the medication is safe and effective. The FDA reviews the application, and decides whether to approve the drug or not.
Drug Labeling: The FDA reviews the labeling / packaging of the drug and ensures that sufficient information is communicated to health care professionals and customers.
Facility inspection: The FDA is inspecting the facilities where the drug is to be made.
Drug approval: The FDA approves the NDA or issues a letter of response.
Post-marketing monitoring: The company is expected to send regular safety reports to the FDA until the FDA approves the medication.
Here we break down three ways in which the FDA keeps you healthy and safe.
1. It ensures your prescription medication is safe and effective
It must have FDA approval before a new prescription medication can be commercialized in the United States. Here’s how it works: First, the drug manufacturer needs to study the drug extensively in various ways, including on humans, to see if it is a safe and successful treatment. The drug manufacturer also needs to fill out an application for FDA approval, and it includes the submission of the test results, the production details and the label to accompany the medication. The label explains how to use the drug as well as potential side effects and hazards. FDA doctors and scientists then review the application and information to ensure that the benefits of the medication outweigh the risks of the medication, according to the FDA web site. And they make sure the drug is produced safely too. If everything checks out, the medication will be approved by the FDA, and the drug company will start selling it.
2. The FDA guarantees that the vaccines are safe for public use
The FDA also controls vaccines, considered to be a biologic product. Vaccines must undergo stringent testing by drug makers according to the FDA criteria. In three phases of clinical trials each vaccine is administered on humans, and thousands of individuals participate. Instead, the FDA must review the findings before authorizing a public-use vaccine. Once it is released to the public, the FDA also monitors the vaccine and its effect. That’s a big deal, again. If the vaccines were not approved by the FDA, drug companies could begin marketing a vaccine that is not effective and/or safe. Rigorous, science-based approval of vaccines has always been critical, but because of the anti-vaccine movement and its ability to wreak havoc on public health, it is particularly necessary right now.
3. The FDA alerts you when new risks arise from a food product or drug
Finally, the FDA is working to inform the public of any shortages of foods and drugs on products they oversee.
The Dental Products Panel (DPP) reviews and examines evidence on the health and efficacy of marketed and testing products for use in dentistry, endodontics or bone physiology in relation to oral and maxillofacial areas and makes relevant recommendations to the Food and Drug Commissioner.
Now that you understand how the FDA operates and what’s the importance of using FDA approved dental materials, you must only order products having FDA approved dental materials in them.
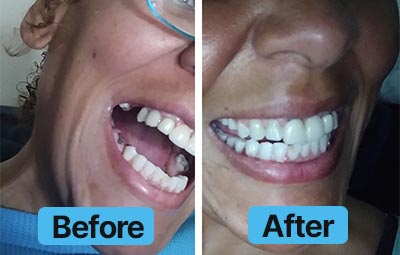
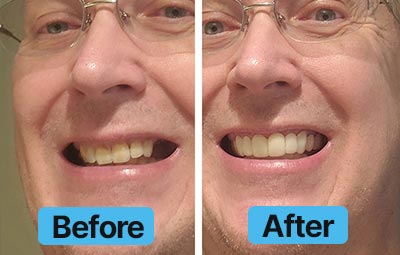
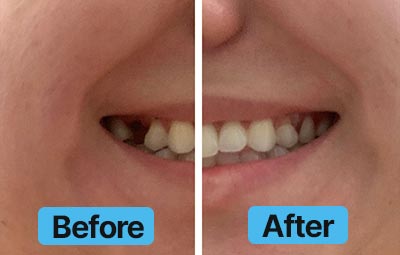
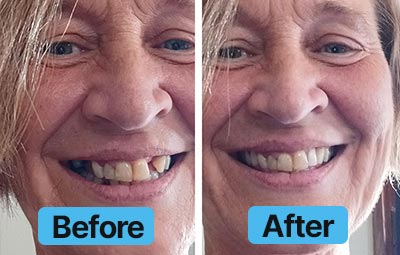
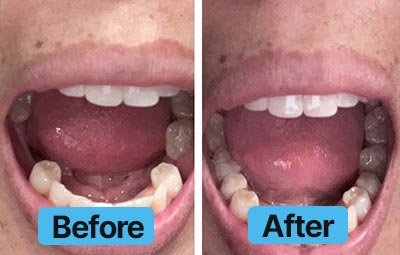
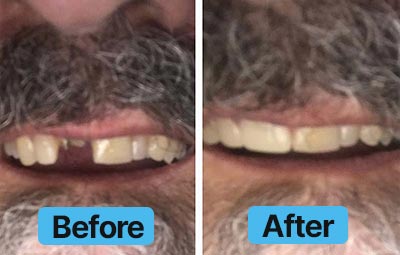
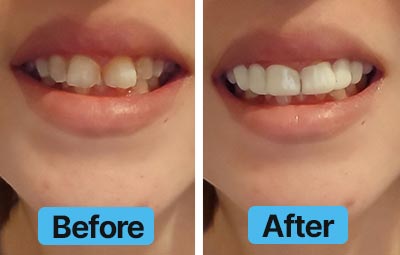
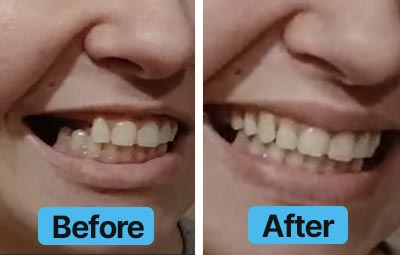
Dental Lab Direct’s all products are made from FDA approved dental materials and handled by certified dental technicians. Our strict quality control and direct customer service ensures you with complete buying confidence. We’re here for you every step of the way!
 60 DAY warranty on all custom-made products | 1,000+ 5 Star ★★★★★ Reviews
60 DAY warranty on all custom-made products | 1,000+ 5 Star ★★★★★ Reviews